Solutions
The main points within the precious metals industry can be divided into three sections: the need for accurate analysis of precious metals, the importance of quality control and compliance with regulations, and the provision of high-precision solutions by Fischer to meet industry requirements. Accurate analysis plays a vital role in determining the value of precious metals, ensuring product quality, and meeting regulatory standards. Fischer’s high-performance equipment and analytical capabilities cater to these needs, providing the necessary precision and quality control for the jewellery and related industries.
The content of gold and other precious metals in an object determines its value, making precise analysis crucial. It is also essential to verify the total composition of the alloy, including metals such as copper and zinc. The main point here is the need for instruments that can provide accurate and speedy material analysis without employing destructive methods. Traditional techniques like Assay, while effective, are time-consuming and destructive, preventing immediate results.
Meeting regulations such as INMETRO Ordinance No. 43/2016, which imposes strict control over harmful elements like cadmium and lead, is crucial. Analyzing precious metals not only helps ensure the quality of products but also assists in cost reduction. The main point here is that precise analysis is paramount to meet regulatory requirements and maintain the safety and health standards associated with the use of precious metals in jewellery and related industries.
The third section highlights the need for high-precision solutions in the jewellery, semi-jewellery, galvanic baths, and pawnbrokers markets. Fischer has developed high-performance equipment and analytical capabilities to meet the industry’s requirements. By offering accurate and reliable solutions, Fischer aims to provide users with the necessary precision and quality control to analyze precious metals effectively. This emphasis on precision and quality is crucial to meet industry standards, comply with regulations, and ensure the value and safety of jewellery and precious metal products.
X-Ray fluorescence solution
Goldscope SD® Series
Tailor-made to analyse gold and precious metals, Fischer’s GOLDSCOPE SD® series offers a solution for the non-destructive testing of materials. The hardware and software of these robust X-ray fluorescence instruments are tailored to the unique requirements of the jewellery and gold industries.
Applications
- Determine the element composition of precious metals such as gold, silver, rhodium, platinum and palladium, as well as less valuable metals such as copper, zinc, etc. The results can be expressed in different units of measurement, such as PPM, percentage, carats and others.
- Measure the thickness of layers for jewellery and semi-jewellery manufacturers, with results expressed in micrometres, micro-inches, among others.
- Analyse galvanic baths (liquids), and measure the chemical composition and contaminants for better-scaling processes (available only with FISCHERSCOPE® XAN).
- Complies with EN 61010, DIN ISO 3497 e ASTM B 568
Markets
- Gold Trading
- Analysis of precious metals
- Manufacture / Import of jewellery and semi-jewellery
Certified Gold Standard
For reliable measurements, it is necessary to use calibration standards that meet the requirements of the quality management
system.
FISCHER offers a wide range of certified standards according to the procedure accredited by DAkkS.
The standard portfolio includes more than 300 calibration standards for EDXRF analysis and covers a large number of applications for
different industries.
Solution analysis using X-Ray fluorescence
Companies need to monitor and control the formulation of their electrolyte baths very closely. Controlling the composition of the bath can determine the quality of the layer applied to the target substrate material. For example, metal coatings of AuCuCd, AuCuIn, RhRu can be monitored both in their composition in the bath and the thickness of the layer applied onto the part.
A particular measuring cell makes it possible to analyse the plating bath and identify any metals in the solution immediately by examining the elements simultaneously in just a few seconds, eliminating the need for sample preparation and use of reagents, for example, in titration.

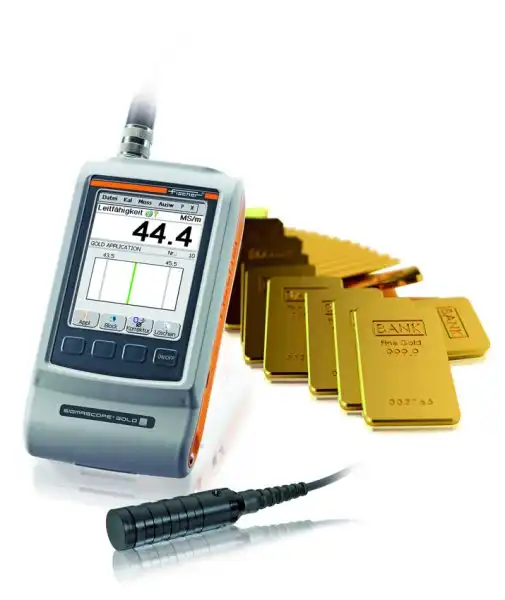
SIGMASCOPE® gold
One of the challenges in the market for buying and selling gold is to ensure authenticity since counterfeit material is often only plated with gold. The SIGMASCOPE® GOLD range is ideal for determining the authenticity of gold using electrical conductivity, analysing the surface and the entirety of the coins, gold bars, and even ingots in a non-destructive, fast and easy way.
Product features of the SIGMASCOPE® GOLD Range
- Reliable determining of electrical conductivity of coins and gold bars
- Non-destructive test method
Measurements in just a few seconds - Positive Material Identification (PMI) of counterfeits
- For testing coins and thin ingots of up to approximately 100 g
- For testing large bars of precious metals from 1 oz (31.1 g) to 1 kg
Contact us now for more information
Whether you know what type of machine you require or have questions surrounding our product range, we are always happy to assist.
Get in touch and we will help you find the Fischer Instrument that best suits your needs. Our team has many years of experience and is always happy to answer your questions.
If you’d prefer to leave your details, please use the form below and we’ll get in touch as soon as we can.